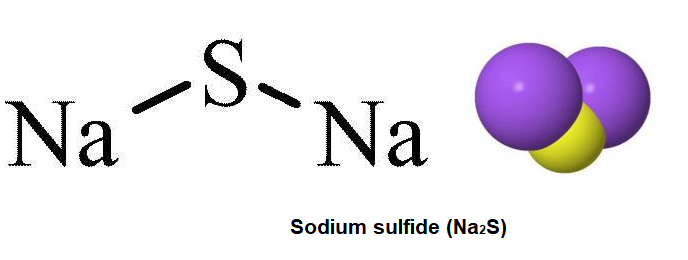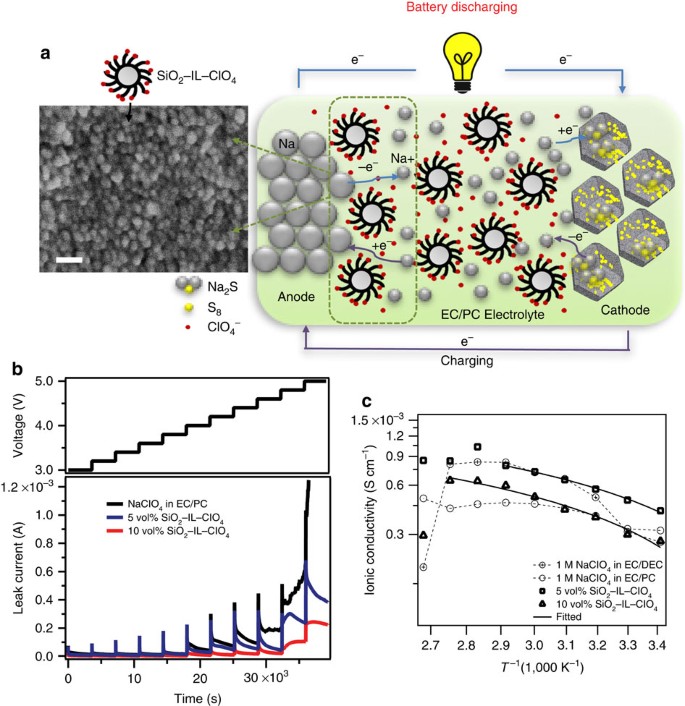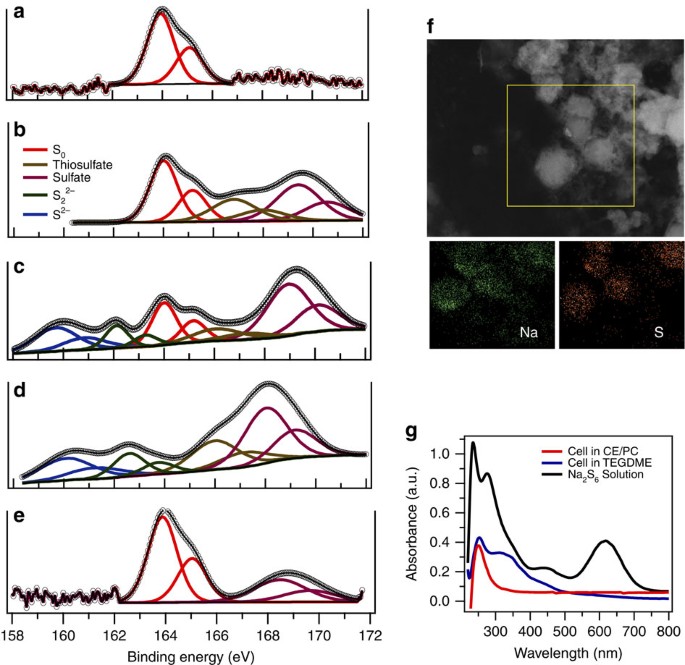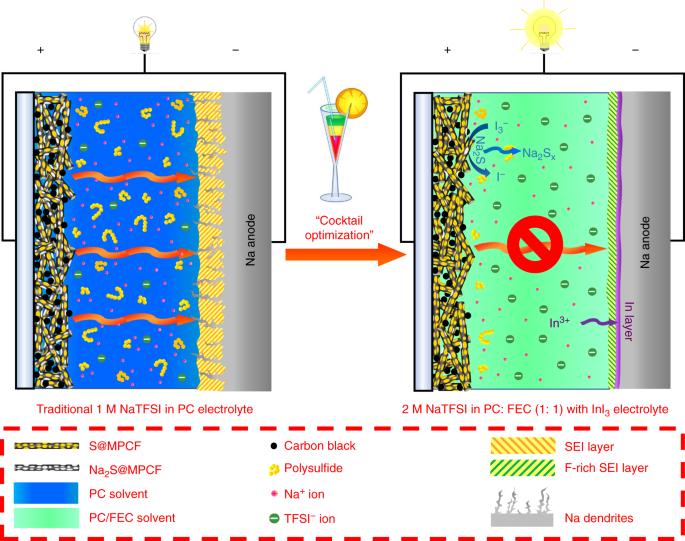
A room-temperature sodium–sulfur battery with high capacity and stable cycling performance | Nature Communications

Najpopularniejsze wyszukiwarki internetowe w Polsce i na Świecie | Blog widzialni.pl - pozycjonowanie i SEO

Unveiling the Advances of 2D Materials for Li/Na-S Batteries Experimentally and Theoretically - ScienceDirect
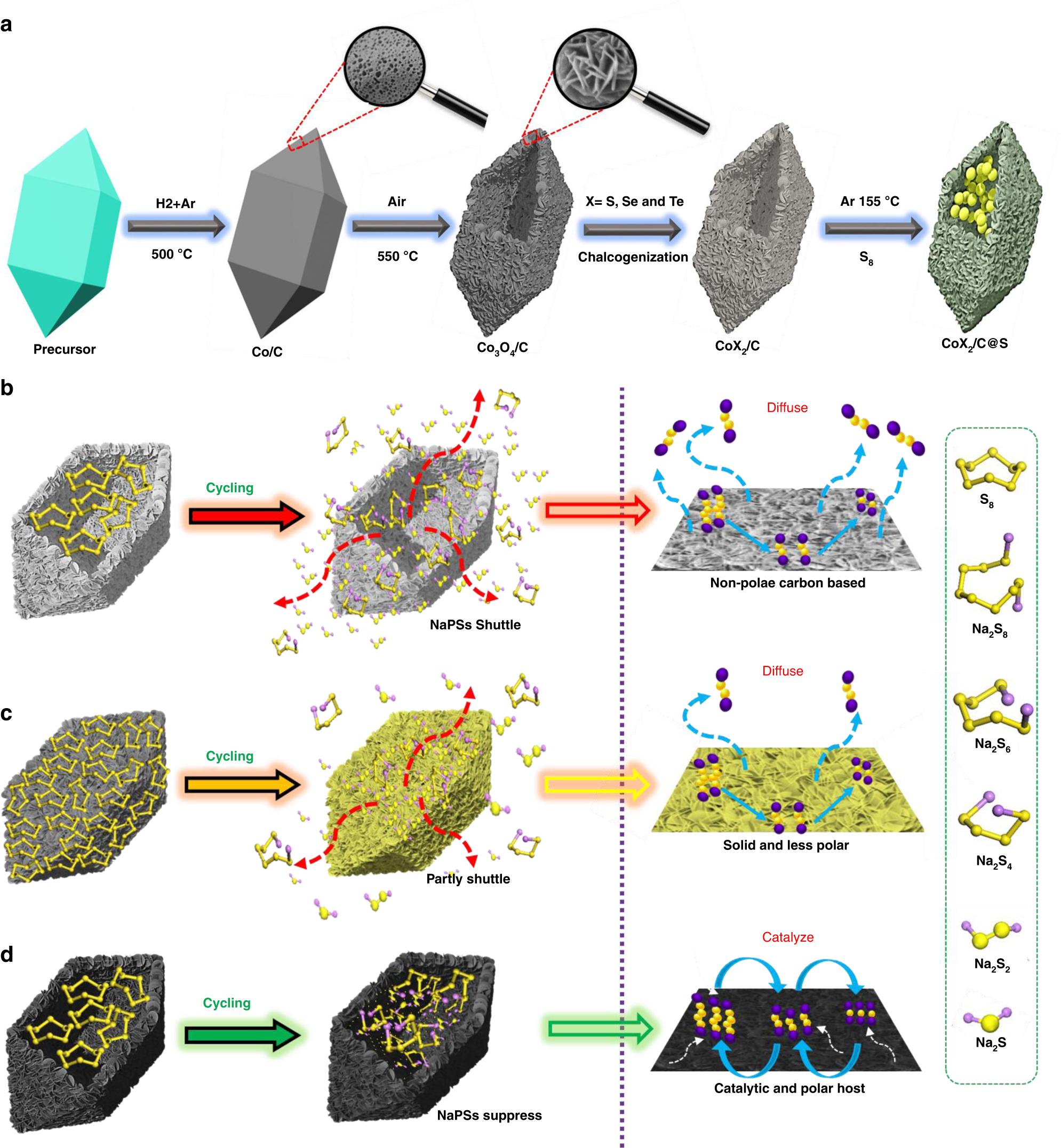
Metal chalcogenide hollow polar bipyramid prisms as efficient sulfur hosts for Na-S batteries | Nature Communications

ENa^ + /Na(s)^0 = - 2.71V , EMg^2 + /Mg(s)^0 = - 2.37V , EFe^2 + /Fe(s)^0 = - 0.44V and ECr^3 + /Cr(s)^0 = - 0.41V Based on this data, state which of the following is the weakest reducing agent.